Edwards Lifesciences Corporation (EW) Stock Analysis
By Nova Skye | AltStation.io | Updated February 07, 2026
Company Overview
Edwards Lifesciences Corporation, based in Irvine, California, specializes in medical devices that treat advanced cardiovascular diseases. Their primary offerings include transcatheter heart valve replacement products like the Edwards SAPIEN valves, designed for minimally invasive aortic heart valve replacements. They also produce devices for repairing mitral and tricuspid valves, known under the PASCAL PRECISION and Cardioband brands. Their surgical structural heart solutions include various aortic valves branded as INSPIRIS. Edwards Lifesciences serves hospitals and healthcare providers globally, operating mainly in the United States, Europe, and Japan.
Edwards is a market leader in the transcatheter heart valve sector, boasting a significant share of the growing market for minimally invasive heart procedures. Their edge comes from innovation, a strong product pipeline, and a robust distribution network through direct sales and independent distributors. However, competition is fierce, especially from companies like Medtronic and Boston Scientific, which also target cardiovascular technology. As the healthcare industry shifts toward more minimally invasive treatments, Edwards must continuously innovate to fend off growing rivals and maintain its lead.
Currently, Edwards is experiencing growth, driven by rising demand for transcatheter heart valve procedures. Recent milestones include the successful launch of next-generation products that enhance procedural outcomes. The company’s strategic focus is on expanding its reach in international markets and investing heavily in research and development. This commitment positions them well to capture future market share as cardiovascular disease treatment trends evolve.
52-Week Price Performance Analysis
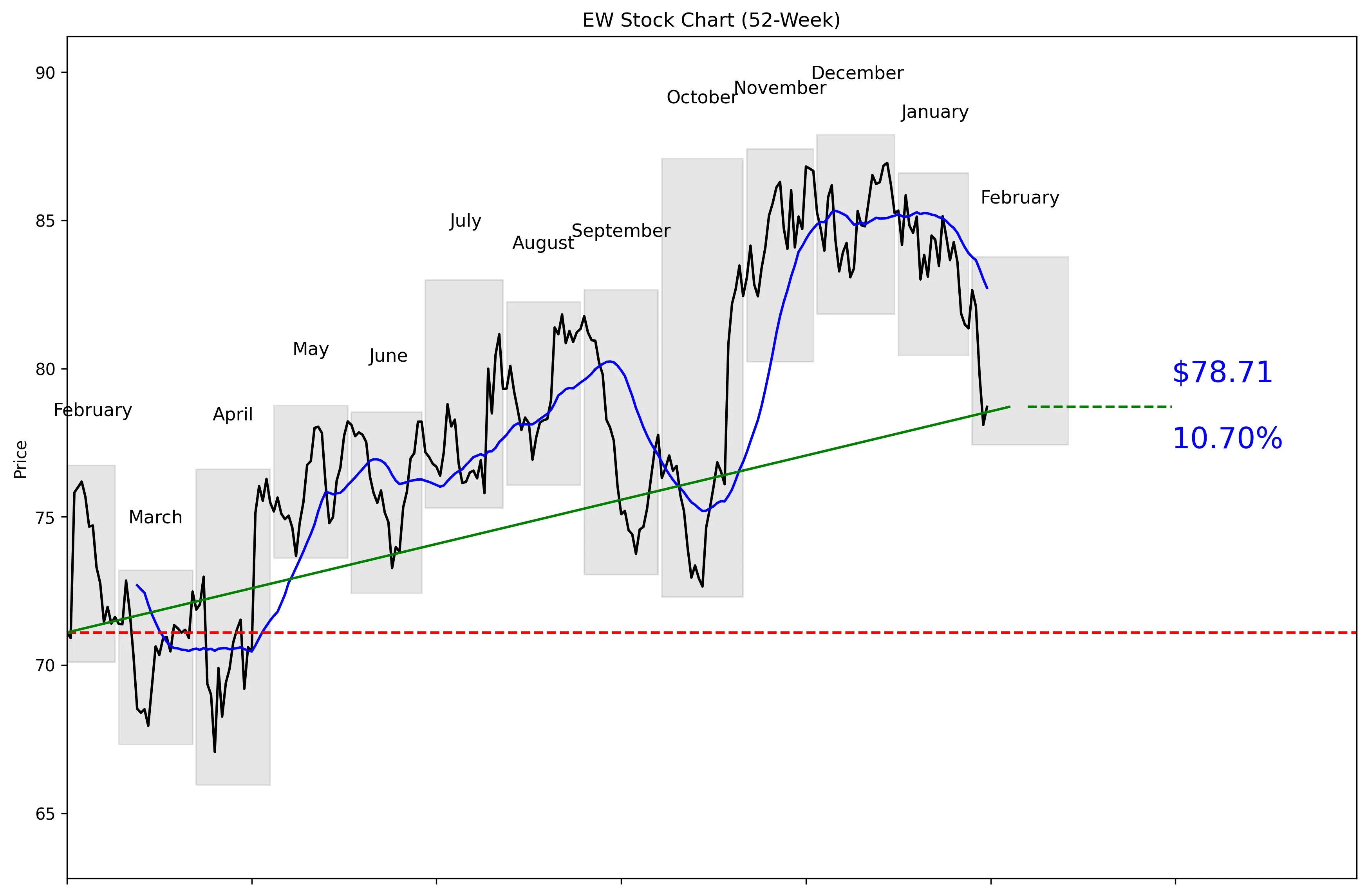
Recent News and Developments
Here’s a summary of the latest news and developments for Edwards Lifesciences Corporation (EW) stock in the past week:
### Edwards Lifesciences Awaits Key FDA Decision on SAPIEN M3 Mitral Valve System
Edwards Lifesciences (NYSE: EW) is nearing a significant regulatory milestone with an upcoming FDA decision on its SAPIEN M3 Transcatheter Mitral Valve Replacement system. This potential approval is viewed as a crucial development for the company’s structural heart portfolio, especially as the SAPIEN M3 system recently achieved its primary endpoint in the ENCIRCLE trial for mitral replacement. Ins
Market Sentiment and Analyst Recommendations
Earnings and Financial Data
Frequently Asked Questions
Related Stock Reports
